Là một dạng thuốc dùng đường uống, viên nang được sử dụng rộng rãi trong các sản phẩm dược phẩm và thực phẩm chức năng. Ở mức độ đơn giản nhất, viên nang là một lớp vỏ bao bọc phần nhân bên trong—thường là bột, hạt hoặc viên nén, và trong một số dạng khác, là chất lỏng hoặc bán rắn.
Hầu hết các quyết định về loại viên nang đều thuộc ba khía cạnh: định dạng (viên nang cứng so với viên nang mềm), vật liệu vỏ (gelatin so với HPMC), và mục tiêu phát hành (hấp thụ tức thì, hấp thụ qua đường ruột, hoặc hấp thụ kéo dài/cải tiến). Khi đã xác định rõ ba yếu tố này, việc thiết kế viên nang phù hợp với nhu cầu ổn định và duy trì sản xuất nhất quán sẽ trở nên dễ dàng hơn nhiều.

Viên nang vẫn được ưa chuộng vì chúng mang lại trải nghiệm sử dụng sạch sẽ đồng thời vẫn đảm bảo tính linh hoạt trong sản xuất dược phẩm và thực phẩm chức năng.
Đối với người dùng, viên nang thường dễ nuốt và có thể giảm thiểu các vấn đề về mùi vị từ một số thành phần. Điều này rất quan trọng đối với các công thức có hương vị mạnh, dư vị kéo dài hoặc các hoạt chất nhạy cảm, nơi trải nghiệm sử dụng dễ chịu hơn sẽ giúp ích.
Đối với các nhà sản xuất, viên nang có thể chứa nhiều loại chất độn hơn so với nhiều dạng thuốc uống khác. Viên nang cứng thường chứa bột, hạt hoặc hệ thống viên nén. Viên nang mềm được sử dụng rộng rãi cho dầu và các chất độn dạng lỏng. Sự linh hoạt này rất hữu ích khi công thức không thể nén tốt thành viên nén, hoặc khi hỗn hợp có thể thay đổi sau này trong khi dạng bào chế vẫn giữ nguyên.
Từ góc độ sản xuất, các sản phẩm dạng viên nang cứng có khả năng mở rộng quy mô hiệu quả vì các bước chiết rót và đóng nắp có thể được tự động hóa trên dây chuyền sản xuất. máy đóng gói viên nang hoàn toàn tự động Điều này giúp duy trì tính lặp lại khi sản lượng tăng lên. Mục tiêu không chỉ là tốc độ mà còn là định lượng nhất quán và xử lý ổn định giữa các lô sản phẩm.
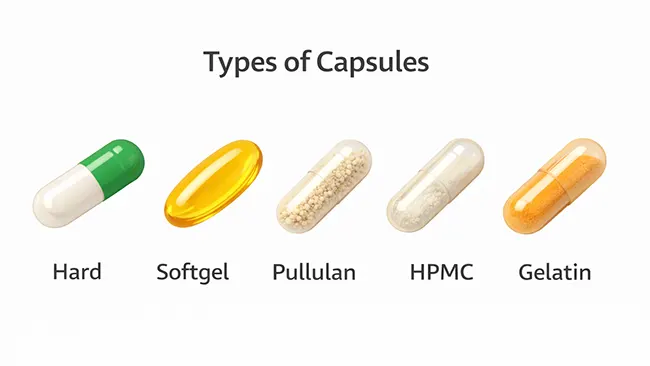
Một cách đơn giản để hiểu các loại viên nang là bắt đầu với hình dạng, sau đó xem xét vật liệu vỏ, rồi làm rõ ý nghĩa của "giải phóng" trong các sản phẩm thực tế.
Viên nang cứng là loại viên nang cổ điển có thiết kế hai phần (nắp và thân). Chúng được sử dụng rộng rãi trong cả thực phẩm chức năng và dược phẩm vì tính linh hoạt và dễ sản xuất ở quy mô lớn.
Bột, hạt hoặc viên/hạt nhỏ thường được sử dụng làm vật liệu độn trong viên nang cứng. Bột thường được dùng cho hỗn hợp và chiết xuất. Hạt được lựa chọn khi cần kiểm soát độ chảy và bụi. Viên/hạt nhỏ thường được sử dụng khi sản phẩm cần có đặc tính giải phóng chậm hoặc được kiểm soát.
Viên nang cứng cũng hỗ trợ nhiều thiết kế sản phẩm “đặc biệt”, chẳng hạn như kết hợp các loại hạt khác nhau trong một liều dùng, hoặc tách các thành phần không nên tương tác trong quá trình bảo quản.
Viên nang mềm là loại viên liền khối và được niêm phong kín. Chúng thường được sử dụng cho dầu, chất lỏng và chất bán rắn, đó là lý do tại sao chúng thường xuất hiện trong các sản phẩm omega-3, dầu vitamin D/K và các công thức gốc dầu khác.
Viên nang mềm thường yêu cầu quy trình sản xuất khác so với viên nang cứng, sử dụng một phương pháp cụ thể. máy đóng gói viên nang mềm Thay vì quy trình đóng gói viên nang cứng. Điểm mấu chốt trong việc lựa chọn "loại" viên nang là sự phù hợp: viên nang mềm được thiết kế cho các chất lỏng bên trong, trong khi viên nang cứng thường được sử dụng cho các chất khô bên trong (với một vài ngoại lệ).
Chất liệu vỏ ảnh hưởng đến khả năng giữ ẩm, độ bền cơ học và thời gian bảo quản thực tế.
Gelatin Nó được sử dụng rộng rãi và hiểu rõ. Nó hoạt động đáng tin cậy khi nhiệt độ và độ ẩm được kiểm soát, nhưng nó có thể phản ứng với sự dao động độ ẩm.
HPMC (hypromellose) Nó thường được định vị là có nguồn gốc thực vật và thường được lựa chọn vì sở thích ăn kiêng hoặc đặc tính giữ ẩm khác biệt. Nó không tự động "tốt hơn" gelatin. Sự lựa chọn đúng đắn là bất kỳ loại vỏ nào giữ được độ ổn định với nhân bên trong, điều kiện bảo quản và thời hạn sử dụng dự kiến.
Hành vi giải phóng chất bên trong viên nang là một nguồn gây nhầm lẫn phổ biến. Vỏ viên nang có thể ảnh hưởng đến tốc độ bắt đầu giải phóng, nhưng nhiều đặc điểm giải phóng được tạo ra bởi thiết kế tổng thể của sản phẩm.
Thông cáo báo chí ngay lập tức Đây là phương pháp tiêu chuẩn: lớp vỏ nhanh chóng bị phá vỡ và phần bên trong được giải phóng.
Đường ruột (giải phóng chậm) Nó được thiết kế để chống lại các điều kiện trong dạ dày và giải phóng chất thải sau đó. Điều này thường đạt được thông qua lớp phủ ruột hoặc thiết kế hệ thống, không chỉ đơn thuần bằng cách chọn một "vỏ bọc đặc biệt" mà thôi.
Việc giải phóng kéo dài/có kiểm soát thường đạt được bằng cách sử dụng các viên/hạt được phủ hoặc các công nghệ giải phóng có kiểm soát khác bên trong viên nang. Hai sản phẩm có thể cùng là "viên nang" nhưng vẫn có hành vi hòa tan rất khác nhau vì thiết kế chất làm đầy khác nhau.
Một số kiểu thiết kế thường xuyên xuất hiện trong sản xuất thương mại vì chúng giải quyết được các vấn đề thực tiễn.
Viên nang cứng chứa chất lỏng sử dụng định dạng viên nang cứng nhưng được làm đầy bằng chất lỏng hoặc bán lỏng, thường có thêm bước niêm phong. Thiết kế viên nang trong viên nang hoặc viên nén trong viên nang tách biệt các thành phần hoặc kết hợp các thành phần trong một đơn vị. Viên nang rắc được thiết kế cho các trường hợp sử dụng mà việc mở viên nang là một phần của quá trình sử dụng.
|
Danh mục viên nang |
Định dạng |
Thông thường đổ đầy |
Ưu điểm chính |
Những điều cần lưu ý thường gặp |
|
Viên nang cứng |
Hai mảnh |
Bột, hạt, viên/hạt nhỏ |
Công thức linh hoạt, có khả năng mở rộng và dễ điều chỉnh. |
Khoảng thời gian tiếp xúc với hơi ẩm; xử lý bụi/tĩnh điện |
|
Viên nang mềm |
Được niêm phong nguyên khối |
Dầu, chất lỏng, chất bán rắn |
Thích hợp nhất cho nhân kem béo; dễ nuốt. |
Khả năng tương thích giữa chất làm đầy và vỏ; kiểm soát nhiệt độ/độ ẩm |
|
Viên nang gelatin |
Cứng hay mềm |
Tùy thuộc vào định dạng |
Nguồn cung dồi dào; hiệu suất đã được chứng minh. |
Mềm đi khi độ ẩm cao; có nguy cơ giòn nếu quá khô. |
|
Viên nang HPMC |
Thường khó |
Bột, hạt, viên/hạt nhỏ |
Định vị dựa trên thực vật; hành vi độ ẩm khác nhau |
Thời gian bảo quản phải phù hợp với lượng chất lỏng và điều kiện khí hậu. |
|
Viên nang ruột |
Cứng hay mềm |
Thường là các đơn vị nạp được phủ lớp |
Phát hành bị trì hoãn |
Cần xác nhận; chiến lược bảo quản và rào chắn rất quan trọng. |
|
Viên nang được sửa đổi/mở rộng |
Thường khó |
Viên nén/hạt/đơn vị siêu nhỏ |
Phát hành có kiểm soát hoặc theo giai đoạn |
Phụ thuộc rất nhiều vào thiết kế đóng gói và kiểm soát quy trình. |

Từ phía người dùng, ý tưởng rất đơn giản: nuốt viên nang, lớp vỏ vỡ ra và chất bên trong được giải phóng. Điều khác biệt giữa các loại viên nang là hình dạng vỏ, cách vỏ hoạt động dưới tác động của độ ẩm/nhiệt độ, và cách thiết kế cơ chế giải phóng chất bên trong.
Về mặt sản xuất, quy trình làm việc khác nhau giữa viên nang cứng và viên nang mềm, nhưng logic vẫn nhất quán: chuẩn bị hỗn hợp ổn định, định lượng chính xác, đóng/niêm phong đáng tin cậy, loại bỏ các khuyết tật, sau đó đóng gói.
Quy trình sản xuất viên nang cứng cấp cao thường trông như thế này:
1.
Chuẩn bị phần nhân
Trộn bột là phương pháp phổ biến. Tạo hạt có thể được sử dụng khi cần kiểm soát độ chảy và bụi. Hệ thống viên nén/hạt nhỏ thường được sử dụng cho các thiết kế giải phóng chậm hoặc có kiểm soát.
2. Chuẩn bị vỏ sò
Vỏ trứng được giữ trong khoảng thời gian cho phép để chúng không bị giòn hoặc quá mềm trong quá trình cho ăn và đóng vỏ.
3. Định hướng và tách nắp/thân máy.
Việc này thường được thực hiện trên máy đóng gói viên nang tự động, giúp ổn định quá trình xử lý và định lượng trong suốt quá trình sản xuất dài.
4. Định lượng chất lỏng với độ đặc vừa phải
Phương pháp định lượng phụ thuộc vào đặc tính của vật liệu cần định lượng và độ đồng nhất mong muốn. Mục tiêu thực tiễn là đạt được trọng lượng vật liệu định lượng lặp lại và sản lượng ổn định.
5. Đóng và khóa (và niêm phong nếu cần)
Các viên nang cứng tiêu chuẩn được đóng kín và khóa chặt. Một số thiết kế có thể cần thêm lớp niêm phong.
6. Loại bỏ bụi và kiểm tra
Các bước thông thường bao gồm đánh bóng/làm sạch bụi vỏ nang và loại bỏ các sản phẩm lỗi trước khi đóng gói.
7. Chuyển sang bao bì
Các viên nang thành phẩm thường được chuyển đến...
máy đóng gói vỉ
hoặc một tuyến đường vận chuyển bằng chai lọ như
dây chuyền đếm viên nang và đóng chai
sau đó đến một
máy đóng thùng
nếu cần thùng carton.
Hầu hết các vấn đề về viên nang xuất phát từ sự không phù hợp giữa vỏ, chất làm đầy và điều kiện bảo quản. Khắc phục chúng thường có nghĩa là điều chỉnh lại hệ thống cho phù hợp, chứ không phải thay đổi loại viên nang một cách mù quáng.
Nhạy cảm với độ ẩm Đây là một thách thức thường gặp. Một số loại vỏ nang có thể bị mềm trong điều kiện độ ẩm cao, trong khi điều kiện quá khô có thể làm tăng nguy cơ giòn. Đó là lý do tại sao các dự án thiết kế viên nang thường xác định sớm thời gian bảo quản và đảm bảo bao bì đáp ứng được khoảng thời gian đó.
Khả năng tương thích với việc đổ đầy Điều này đặc biệt quan trọng đối với các loại viên nang mềm và viên nang chứa chất lỏng. Một số loại dầu, dung môi hoặc thành phần phản ứng có thể gây áp lực lên vỏ viên nang theo thời gian. Việc kiểm tra tính tương thích ở đây giúp ngăn ngừa rò rỉ chậm, vỏ viên nang yếu đi hoặc thay đổi hình thức trong suốt thời hạn sử dụng.
Hành vi dòng chảy và bụi Ảnh hưởng đến tính nhất quán trong định lượng. Bột xốp, dễ bị tĩnh điện hoặc có độ chảy không ổn định có thể gây khó khăn trong việc kiểm soát trọng lượng. Phương pháp tạo hạt hoặc viên thường cải thiện khả năng xử lý, nhưng chúng cũng có thể làm thay đổi hành vi giải phóng sản phẩm và các yêu cầu xác nhận.
Việc lựa chọn giữa các loại viên nang trở nên dễ dàng hơn khi quyết định dựa trên hình thức, chất liệu và mục tiêu phát hành.
Viên nang cứng là lựa chọn mặc định tốt cho các sản phẩm dạng bột, hạt và viên nén, và chúng dễ dàng mở rộng quy mô thông qua hệ thống đóng gói và đóng nắp tự động. Viên nang mềm thường phù hợp nhất với các loại dầu và chất lỏng, miễn là khả năng tương thích giữa chất làm đầy và vỏ viên nang được xác nhận.
Cơ chế giải phóng thuốc cần được làm rõ hơn. Giải phóng tức thì là phổ biến và đơn giản. Các sản phẩm giải phóng có kiểm soát và giải phóng kéo dài thường được tạo ra thông qua lớp phủ hoặc công nghệ đóng gói (thường là hệ thống viên/hạt) chứ không chỉ dựa vào lớp vỏ tiêu chuẩn.
Nếu định dạng viên nang phù hợp với lượng thuốc bên trong, vật liệu vỏ phù hợp với thời gian bảo quản và thiết kế cơ chế giải phóng thuốc phù hợp với mục tiêu sản phẩm, thì kết quả thường là sản phẩm ổn định, có khả năng mở rộng quy mô và dễ dàng đóng gói một cách nhất quán.
Các dạng bào chế chính là viên nang cứng (hai mảnh) và viên nang mềm (một mảnh được niêm phong). Nhiều danh sách cũng phân loại theo chất liệu vỏ (gelatin so với HPMC) và theo đặc tính giải phóng thuốc (giải phóng tức thì, giải phóng qua đường ruột, giải phóng kéo dài/có tác dụng điều chỉnh).
Viên nang cứng thường được sử dụng cho các loại bột, hạt và viên nén. Viên nang mềm thường được sử dụng cho dầu, chất lỏng và chất bán rắn. Sự lựa chọn tốt hơn phụ thuộc vào dạng đóng gói, nhu cầu về độ ổn định và khả năng tương thích.
Gelatin được sử dụng rộng rãi và hoạt động tốt trong điều kiện được kiểm soát. HPMC thường được lựa chọn cho các sản phẩm có nguồn gốc thực vật hoặc các đặc tính về độ ẩm khác nhau. Lựa chọn tốt nhất là loại giữ được độ ổn định với chất làm đầy và điều kiện bảo quản của bạn.
Thuật ngữ “enteric” thường có nghĩa là giải phóng chậm – được thiết kế để chống lại các điều kiện trong dạ dày và giải phóng thuốc sau đó. Điều này thường đạt được thông qua các lớp phủ hoặc thiết kế hệ thống chứ không chỉ đơn thuần là lớp vỏ thông thường.
Thông thường, khả năng giải phóng có kiểm soát được tạo ra bởi thiết kế chất làm đầy (ví dụ: viên/hạt được phủ lớp) chứ không chỉ bởi lớp vỏ tiêu chuẩn.
Có. Viên nang cứng chứa chất lỏng có tồn tại và thường cần bước niêm phong để đảm bảo tính toàn vẹn và ổn định lâu dài.
Vỏ nang nhạy cảm với độ ẩm và nhiệt độ. Quá khô có thể làm tăng nguy cơ giòn; quá ẩm có thể làm mềm vỏ và ảnh hưởng đến việc xử lý. Xác định thời gian bảo quản và chiến lược đóng gói phù hợp thường giải quyết được vấn đề này.
● ICH Q1A(R2) Thử nghiệm độ ổn định của dược chất và sản phẩm mới:
https://database.ich.org/sites/default/files/Q1A%28R2%29%20Guideline.pdf
● Cổng thông tin hướng dẫn của FDA (tìm kiếm dạng bào chế rắn uống giải phóng chậm / SUPAC-MR):
https://www.fda.gov/regulatory-information/search-fda-guidance-documents